
Phofo ea Cobalt Oxide e Ntšo ea Co3O4 Phofo
Tlhaloso ea Sehlahisoa
Cobalt tetroxide phofo ke phofo e ntšo, e ke keng ea qhibiliha ka metsing, e nang le metallic luster le conductivity e ntle ea motlakase.Cobalt tetroxide e na le oxidizing haholo 'me e ka lokolla oksijene sebakeng se nang le asiti.Cobalt tetroxide phofo ke mohloli oa bohlokoa, o ka sebelisoang ho kopanya ammonia, formaldehyde, oxalic acid le metsoako e meng ea manyolo.Hape ke lisebelisoa tse phahameng tsa makenete le pigment.Ha ho etsoa betri, hangata e sebelisoa e le lisebelisoa tse ntle tsa electrode, tse ka fokotsang ka katleho ho hanyetsa ka hare ho betri.Ho phaella moo, e ka sebelisoa e le selelekela bakeng sa lubricant le lisebelisoa tse phahameng tsa mocheso o phahameng oa mocheso.
Tlhaloso
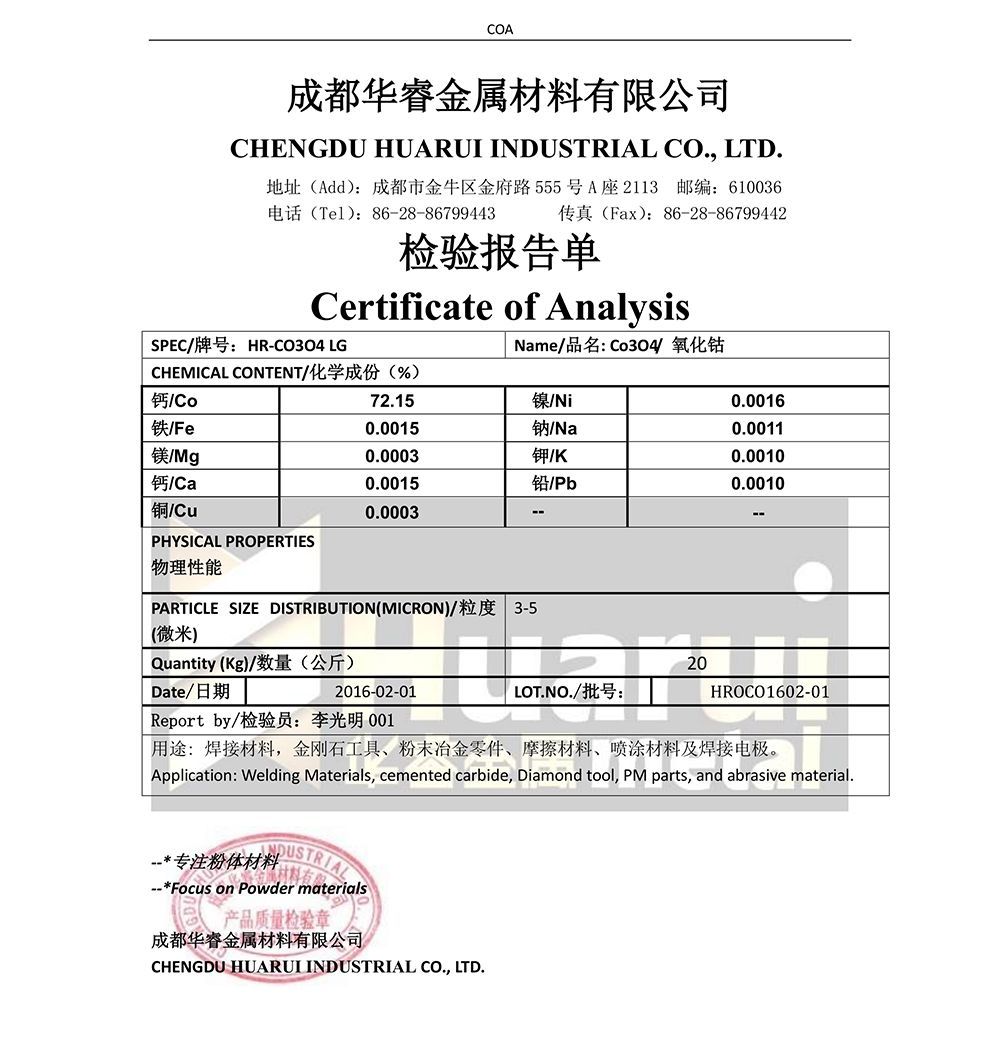
| Sebopeho sa phofo ea Cobalt oxide | ||||||
| Kereiti | Ho se hloeke ho na le (wt% max) | |||||
| Co% | Ni% | Cu% | Mn% | Zn% | Fe% | |
| A | 73.5±0.5 | ≤0.05 | ≤0.003 | ≤0.005 | ≤0.005 | ≤0.01 |
| B | ≥74.0 | ≤0.05 | ≤0.05 | ≤0.05 | ≤0.05 | ≤0.1 |
| C | ≥72.0 | ≤0.15 | ≤0.10 | ≤0.10 | ≤0.10 | ≤0.2 |
Koa
Kopo
1. E sebelisoa e le colorant le pigment bakeng sa khalase le lirafshoa, motsoako o thata;
2. Li-oxidants le li-catalysts indastering ea lik'hemik'hale;
3. E sebelisoa indastering ea semiconductor, li-ceramics tsa elektronike, lisebelisoa tsa cathode tsa lithium ion, lisebelisoa tsa makenete, mocheso le lisele tsa khase;
4. E sebelisoa e le reagent e phahameng ea ho hloeka analytical, cobalt oxide le ho lokisetsa letsoai la cobalt
Mokhoa oa ho laola boleng

Huarui e na le sistimi e thata ea taolo ea boleng.Re leka lihlahisoa tsa rona pele ka mor'a hore re qete tlhahiso ea rona, 'me re leka hape pele ho thomello e' ngoe le e 'ngoe, esita le sampole.'Me haeba u hloka, re ka thabela ho amohela motho oa boraro ho etsa tlhahlobo.Ehlile haeba u rata, re ka u fa sampole hore u e hlahlobe.
Boleng ba sehlahisoa sa rona bo netefalitsoe ke Sichuan Metallurgical Institute le Guangzhou Institute of Metal Research.Tšebelisano ea nako e telele le bona e ka boloka nako e ngata ea liteko bakeng sa bareki.











